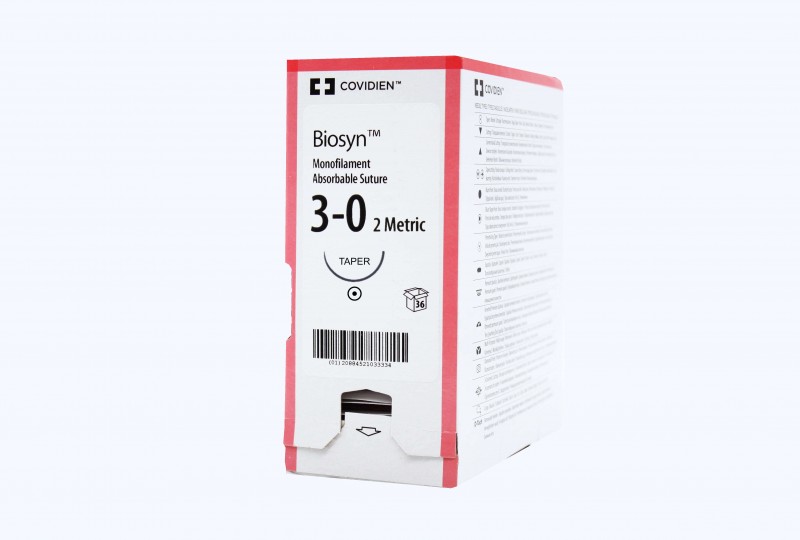
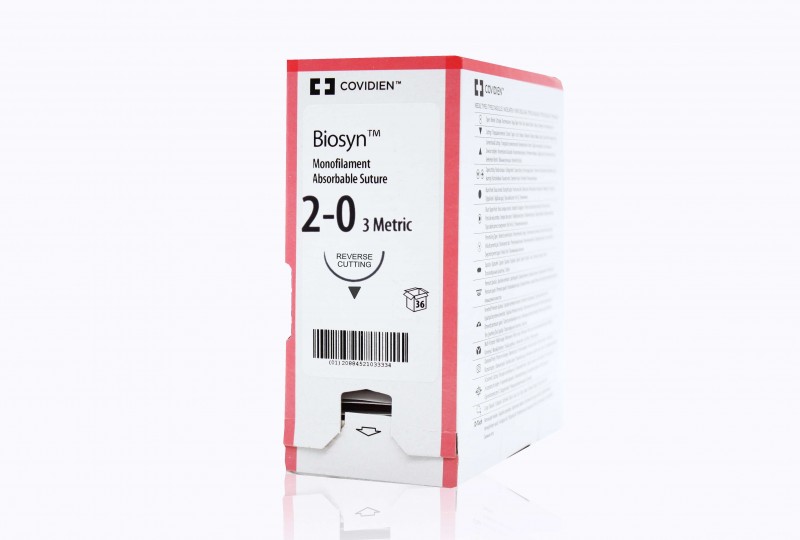
LSI Solutions TK Ti-Knot Device
In Stock: 50 Eaches
Express: Need it tomorrow? Order in next 2 hours 8 minutes and choose overnight shipping at checkout.
The item listed above is expired and may be used for educational, training, and non-clinical research purposes only. Any product information appearing below, including the product indication statement, pertains to an in-date item only.
Additional information
| Size | 1 |
|---|---|
| Arms | N/A |
| Manufacturer | |
| Type | Ti-KNOT |
| Unit | box of 1 |
- FDA Product Code: HCF
- FDA Product Code Name: Instrument, Ligature Passing And Knot Tying
- GMDN Term Code: 61353
- GMDN Term Name: Suture clasp, non-bioabsorbable
- GMDN Term Description: A small, sterile, implantable fastening device designed to secure the ends of a suture as an alternative to tying a knot, for approximation of soft tissue and/or prosthetic materials during a surgical procedure. It is made of non-bioabsorbable materials [e.g., titanium (Ti)] and is typically implanted with a dedicated applicator.
LSI
LSI Solutions is a leading medical device company dedicated to advancing minimally invasive surgical procedures through innovative technologies. Based in Victor, New York, LSI Solutions focuses on developing and manufacturing cutting-edge devices for cardiothoracic and general surgery. The company’s portfolio includes precision-crafted suturing systems, surgical instruments, and accessories designed to improve procedural efficiency and patient outcomes. With a strong commitment to surgeon collaboration, quality, and innovation, LSI Solutions continues to pioneer advancements in surgical care, empowering healthcare professionals to deliver safer, less invasive, and more effective treatments.