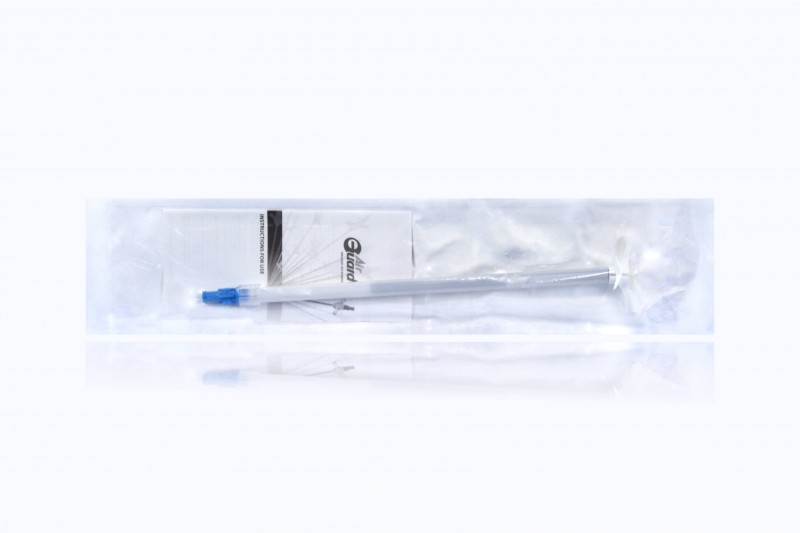
Bard AirGuard Valved Introducer 10F x 15cm
In Stock: 41 Eaches
Express: Need it tomorrow? Order in next 2 hours 8 minutes and choose overnight shipping at checkout.
The item listed above is expired and may be used for educational, training, and non-clinical research purposes only. Any product information appearing below, including the product indication statement, pertains to an in-date item only.
Additional information
| Size | 10.0F |
|---|---|
| Arms | N/A |
| Manufacturer | |
| Type | Introducers |
| Unit | eaches |
- FDA Product Code: DYB
- FDA Product Code Name: INTRODUCER, CATHETER
- GMDN Term Code: 58865
- GMDN Term Name: Vascular catheter introduction set, nonimplantable
- GMDN Term Description: A collection of nonimplantable, invasive devices intended to provide percutaneous vascular access to enable introduction of a catheter (not included) into the vascular system. It includes a non-steerable introducer sheath with an inner obturator/dilator, and typically an additional access device(s) [e.g., guidewire, introducer needle, syringe, additional dilators] which may include a haemostatic valve to control blood loss, usually to enable arterial access. This is a single-use device.
Bard PV
C.R. Bard (a part of BD - Becton, Dickinson and Company) was a global leader in the development and manufacturing of innovative medical technologies. Known for their high-quality products, Bard specialized in solutions for vascular access, urology, oncology, and surgical specialties. Their product portfolio included catheters, biopsy devices, implantable ports, and hernia repair systems, designed to enhance patient care and procedural efficiency. As part of BD, the legacy of C.R. Bard continues to drive advancements in healthcare, delivering trusted solutions that improve outcomes for patients and providers worldwide.